News and Announcements
Drs Elman and Schechet Participate in FDA Study
Dr. Elman and Dr. Schechet were part of a cutting-edge wet AMD research study to validate the usability and visualization performance of the new first-of-its-kind Home OCT system leading to FDA marketing authorization. This technology is used during virtually every visit for a patient with wet AMD, and having it in the patient’s home to use daily will significantly improve patient care and lead toward better outcomes.
Michael J. Elman, M.D. presented trial results at the 48th Annual Meeting of the Macula Society

Dr. Schechet interviewed on the “Open Your Eyes” Podcast.
Open in Apple Podcasts
Elman Retina’s own Sidnet Schechet, MD was featured on the Open Your Eyes Radio Show
Subscribe: Spotify
Get ready for an eye-opening conversation this week on Open Your Eyes Radio! Dr. Kerry Gelb welcomes renowned expert Dr. Sid Schechet to explore the fascinating connection between your overall health and your body’s peak performance as you age. Discover practical insights to help you stay strong, sharp, and thriving at every stage of life!
Dr. Schechet’s Background: Dr. Schechet graduated Magna Cum Laude from Yeshiva University. He earned his medical degree from Baylor College of Medicine, one of the top 20 medical schools in the country. While at Baylor, Dr. Schechet served as the president of the ophthalmology club. Following an internship year at Medstar Harbor Hospital in Baltimore, Maryland, Dr. Schechet completed his ophthalmology residency at the University of Maryland, where he served as the chief resident during his senior year of training. During his ophthalmology trainings, he received the “Outstanding Resident” award at the Greater Baltimore Medical Center and was awarded the “Gold VA Pin Award” by the director of the Maryland Veterans Administration (VA) healthcare system. Dr. Schechet next completed a rigorous and prestigious two-year retina fellowship at The University of Chicago, where he performed cutting-edge research on clinical and surgical retinal treatments.
Dr. Schechet is board-certified by the American Board of Ophthalmology. He has authored multiple research articles in leading ophthalmology journals, such as “RETINA,” as well as retinal book chapters including the renowned Ryan’s Retina textbook. Dr. Schechet has presented his research findings and has won multiple awards for videos of his innovative surgical procedures at premier, international conferences including the American Academy of Ophthalmology Annual Conference; The Macula Society; The American Society of Retina Specialists; and the Vit-Buckle Society. He is an active member of, The American Academy of Ophthalmology, the American Society of Retina Specialists and American Retinal Forum. Dr. Schechet considers it a privilege and his professional responsibility to contribute to these organizations by diligent participation, research, and academic writing.
Dr. Schechet is a devoted husband and father who enjoys playing sports and hiking with his family.
Enjoy the show!
We are now open in Catonsville!


Foundation Fighting Blindness presents Wizard of the Eyes at the Visionwalk 2024

Don’t Get Burned by the 2024 Great American Eclipse


Michael J Elman, M.D. presented the results of the Clinical Utility of Home OCT Data in Management of Neovascular Age Related Macular Degeneration at the 47th annual meeting of the Macula Society in Palm Springs, California on February 9, 2024. Home OCT offers the hope of improving outcomes in the treatment of retinal diseases by allowing treating physicians to personalize treatment specific to that patient’s response. Dr. Elman has been actively involved in developing Home OCT with Notal Vision based in Tel Aviv, Israel. Previously, Dr Elman was a Principal Investigator on the National Eye Institute supported HOME Trial that established the superior role of Notal’s Foresee Home Monitoring System in preserving vision in eyes at risk for neovascular AMD. Elman Retina’s Dr. Sid Schechet currently serves as Principal Investigator in the DRCR Retina Network’s AO clinical trial comparing Home OCT eyes with standard of care in the treatment of neovascular AMD eyes.
Dr. Elman Achieves a Remarkable Milestone with Over 7700 Citations
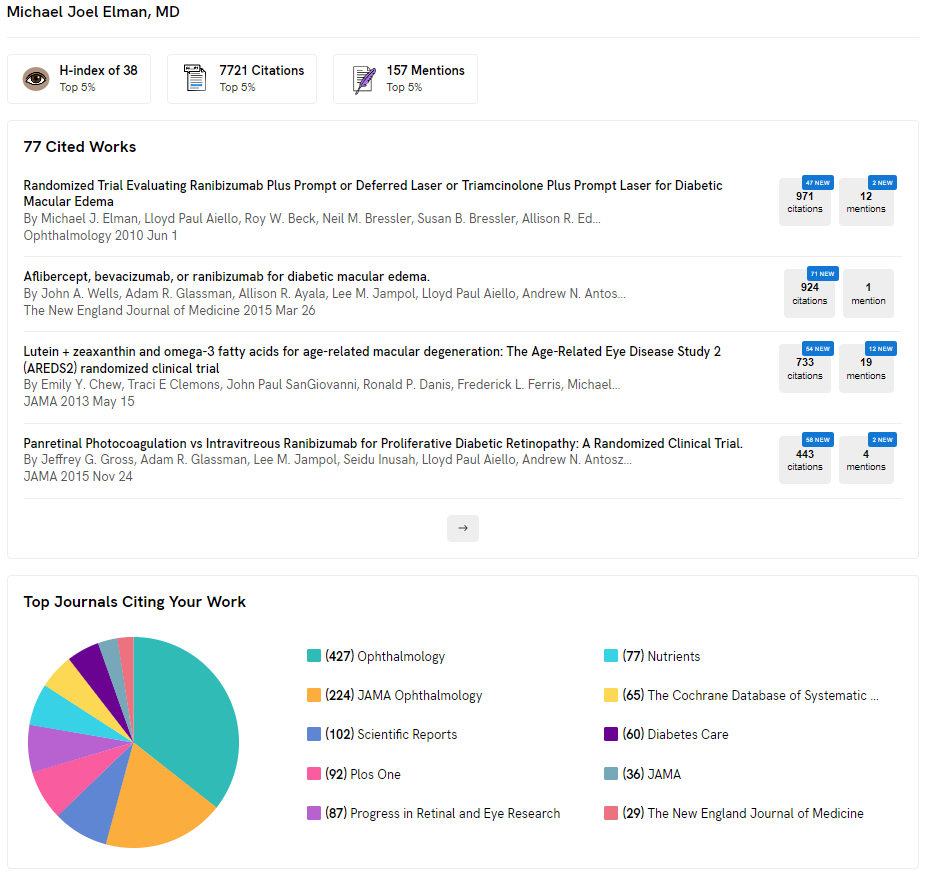
In an extraordinary feat, Dr. Elman has reached an impressive milestone by accumulating over 7700 citations. This significant achievement positions Dr. Elman among the top 5% of researchers in terms of citations within the esteemed Doximity community!
Elman Retina is proud to help pioneer the future of predictive analytics with digital healthcare. A recently published sub-analysis of ALOFT study shows how #remotemonitoring can generate aggregates of large data sets and provide unique insights, specifically the predictive nature of non-exudative ForeseeHome alerts.
Reviewing data from more than 3,000 eyes monitored on the ForeseeHome program, the report looked at the future rates of conversion to wet #AMD. Patients receiving a non-exudative alert were nearly 2x more likely to convert in the next year compared to the baseline population. This finding can be very meaningful for the management of these high-risk patients!
Dr. Elman presenting the at Macula Society Annual Meeting



Read the full publication here.
Pneumatic Retinopexy in Dialysis-Associated Rhegmatogenous Retinal Detachments
In this retrospective case series, six eyes in six patients with dialysis-associated retinal detachments were successfully repaired with one pneumatic retinopexy procedure. Read more.
Ultrasound Imaging of Extensive Peripheral Macrocysts in Chronic Total Combined Rhegmatogenous-Traction Retinal Detachment
Working with Eric, a stellar medical student at the University of Maryland, Dr. Schechet recently had another research publication in the prestigious American Journal of Ophthalmology Case Reports titled: “Ultrasound Imaging of Extensive Peripheral Macrocysts in Chronic Total Combined Rhegmatogenous-Traction Retinal Detachment”
Discussion. Uncontrolled proliferative diabetic retinopathy (PDR) can progress to serious ocular complications such as TRD or in this case, a combined rhegmatogenous-traction retinal detachment.
Read the full publication here.
Diabetic Retinopathy in Pregnancy
Dr. Schechet was invited to speak about an important topic at the Johns Hopkins Wilmer Glaucoma Division’s Fresh Air Meeting – “Ergonomics for Ophthalmologists”

Suprachoroidal Hemorrhages During Cataract Surgery
Is surgical intervention the only treatment option?
BY WEI CHEN LAI, BS; MICHAEL J. ELMAN, MD; AND SIDNEY A. SCHECHET, MD
Staff Appreciation!
The Elman Retina Group team enjoyed a surprise ice cream truck party to show our sincere appreciation for all the hard work that every single team member puts in every single day. It’s been a rough year for society due to COVID, but we are so proud of our ERG family for pushing through the tough times and keeping the top-notch work ethic, happiness, and empathy that makes the Elman Retina Group stand out from the rest. THANKS FOR YOUR HARD WORK AND DEDICATION!











Comprehensive Patient Monitoring For AMD
Dr. Sidney A. Schechet
A review of how evolutions in digital health allow AMD patients to receive comprehensive monitoring services while at home.
We’re proud to announce that Dr. Elman was just voted #1 in the Vision Care section of the 2021 Baltimore Jewish Times’ annual “Best of Jewish Baltimore” competition.
Retinoblastoma presenting as pseudohypopyon and preserved visual acuity.
Purpose:
To describe a Case of retinoblastoma that presented subtly as a pseudohypopyon in a child with preserved visual acuity.
Macular Hole Closure with Medical Treatment
Dr. Schechet and his team just published new research regarding macular hole closure with medical treatment.
Survey of musculoskeletal disorders among US ophthalmologists
Dr. Schechet and his team just published new research showing high rates of musculoskeletal disorders among practicing ophthalmologists. This is important for eye surgeons to know and prevent these problems with proper ergonomic behaviors.

Syphilitic retinitis presentations: punctate inner retinitis and posterior placoid chorioretinitis, published in the National Library of Medicine by Dr. Schechet and his co-authors. Click Here
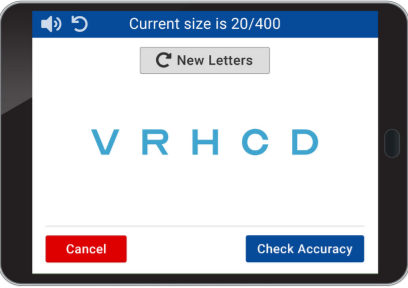
‘Use of the King-Devick Variable Color Contrast Sensitivity Chart to differentiate stages of age-related macular degeneration,’ published in BMJ Open Ophthalmology by Dr. Schechet and his co-authors.
‘Bilateral retinal detachments presenting as manifestations of Vogt-Koyanagi-Harada syndrome,’ published in European Journal of Rheumatology by Dr. Schechet and his co-authors.
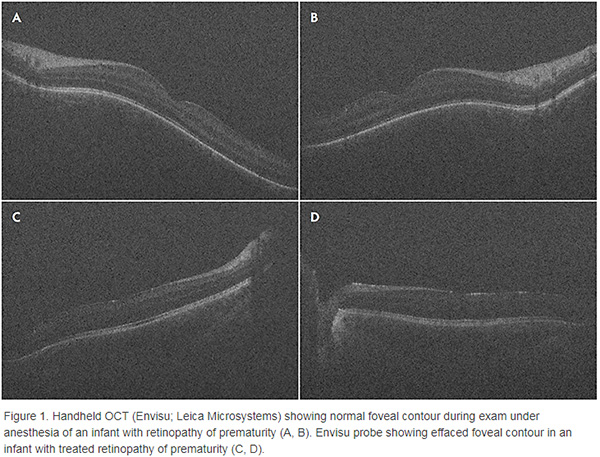
Update on Intraoperative OCT for Vitreoretinal Surgery
A new technology impacting surgical technique, management, and decision-making.
By SIDNEY A. SCHECHET, MD • MICHAEL J. ELMAN, MD • RAHUL KOMATI, MD • MICHAEL P. BLAIR, MD • SEENU M. HARIPRASAD, MD
January 1, 2020

Dr. Schechet presented an interesting surgical case and discussed the latest advancements in complex macular hole repair at the GBMC Ophthalmology Department’s CME evening event.

Dr. Schechet was in London for the 52nd international Retina Society meeting (http://www.retinasociety.org/). Here he is next to his research poster, in which he presented “Bevacizumab or Laser for Aggressive Posterior Retinopathy of Prematurity.” This study was recently published in a major peer-reviewed journal: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6302568
Outcomes of vitrectomy for diabetic tractional retinal detachment
Contralateral amaurosis after a retrobulbar block

On June 26, 2019, Michael J. Elman, M.D. was the guest of Professor Eric Souied, as Dr. Elman visited the world famous Ophthalmology Department at the University of Paris, Creitil. Here Dr. Elman (right) is pictured with Dr. Souied (left), Chief of Ophthalmology, and Dr. Alexandra Miere (center). During his visit, Dr. Elman was shown ground breaking methods for analyzing OCT angiograms, which Dr. Elman will now be able to use to aid in early diagnosis of macular diseases, such as age related macular degeneration, diabetic retinopathy and retinal vascular occlusion. Dr. Elman is among the first in the United States to use this revolutionary diagnostic technology developed by Dr. Souied and his team in France.
Michael J. Elman, M.D. was invited to serve as a moderator at the diabetes session of the biennial international retina meeting, Maculart 2019, in Paris on June 24, 2019. In addition, Dr. Elman presented the results of the “Exploratory Analysis of Change in Visual Fields Over 5 Years Among Eyes with Proliferative Diabetic Retinopathy” on behalf of the DRCR Retina Network.


Itay Kazaz and Dr. Michael Elman presenting research from the Elman Retina Group at the annual international ARVO meeting in Vancouver, May 2, 2019
April 23, 2019